Welcome to the

The Laboratory of Neuroscience has several ongoing projects, involving three major areas: Aptamers, Stem Cells and Neuroscience.
Furthermore, many projects are intended to elucidate the involvement of the purinergic and kininergic systems in diverse physiological and pathological processes, for example, in celular differentiation, in neurodegenerative diseases and in cancer cells.

The registration for the XII Meeting of the Brazilian Purines Club is open!
Come sign up!

Job offer for post-docs
We are looking for two highly motivated people to participate as post-doctoral fellows within scientific projects at the Warsaw Medical University at the Department of Regenerative Medicine.
Supervisors: Prof. Mariusz Ratajczak (Medical University of Warsaw) in collaboration with Prof. Henning Ulrich (Institute of Chemistry, University of São Paulo)
Details:
Type of employment relationship: Contact of mandate
Employing entity: Warsaw Medical University
Expected start date: could be immediately
Duration: 36-month position Salary: 10 000 PLN (tax included)
Projects description:
- Early and late consequences of stem cell compartment damage due to SARS-CoV-2 infection
COVID19 or SARS-CoV-2 virus is single-stranded RNA virus, whose infection can be asymptomatic or lead to damage of several vital organs and a fatal complication involving “cytokine storm”, which results in uncontrolled hyperactivation of the immune response by innate immunity cells. The major concern is that we still cannot foresee late complications of this infection including direct or indirect effects on stem cell compartment. SARS-CoV-2 may enter human cells after binding to the angiotensin- converting enzyme 2 (ACE2) receptor and utilizes its surface spike protein (SP) for attachment and entry into the target cells. It has been demonstrated that ACE2 receptor is highly expressed on hematopoietic stem cells (HSCs) and endothelial progenitors (EPCs) isolated from adult hematopoietic organs as well on developmental early precursors of these cells. Its expression increases with more primitive phenotype of cells and it may explain that our group noticed its high expression in addition to HSC and EPC on human very small CD133+CD34+Lin-CD45– cells, which can be specified as reported by us and others into functional HSCs and EPCs. COVID19 after binding to ACE2 may hyperactivate Nlrp3 inflammasome as we recently demonstrated in cells at different level of specification into hematopoietic and endothelial lineage. This may lead to pyroptotic death of the cells exposed to virus SP. Moreover, this could lead also as we postulated of an initiation of “cytokine storm” by innate immunity cells. Evidence accumulates that COVID19 infection despite a fact that it manifests primarily as a respiratory syndrome has significant impact on other organs including the hematopoietic system and endothelium leading to several complications. To support this a large percentage of infected patients, suffer from lymphopenia and thrombocytopenia as well as from damage of endothelium that promotes hypercoagulability. Nevertheless, there are still not very well known mechanisms how virus affect human stem cells and damage them by productive or abortive infection. It is well known that the innate immune response and activation of the Nlrp3 inflammasome are important defense mechanisms during the first days of infection, until acquired immunity responds with the production of antibodies. However, as mentioned above hyperactivation of this intracellular protein complex in innate cells may induce a cytokine storm or may lead to their death of other cells in mechanism of pyroptosis. Virus may also damage cells by lysis or theoretically what we hypothesize may stay after entry into long living stem cells in a latent form and become activated when immune system becomes impaired. Our group postulated a possibility that damage of stem cells for hemato/endothelial lineage may occur mainly by hyperactivation of Nlrp3 inflammasome after binding of viral SP to ACE2 expressed on these cells. Similar role may play interaction of SP with Toll like receptor-4 (TLR4). Our group and group of Dr. Hal Broxmeyer has demonstrated that exposure of umbilical cord blood-derived HSCs to SP protein decreases viability and in vitro clonogenicity of these cells. We also observed similar effect on proliferation of human EPC. Based on this a central hypothesis of our proposal is that COVID19 infection may damage by SP-ACE2 or SP-TLR4 interaction stem cells from hematopoietic/endothelial lineage which contributes to early and late consequences of this infection.
- Role of Nox2-ROS-Nlrp3 inflammasome axis in homing ang engraftment of hematopoietic stem/progenitor cells
Delayed engraftment of hematopoietic stem/progenitor cells (HSPCs), or even failure to engraft, is still a significant clinical problem, mainly if the number of HSPCs is limited. The mechanisms that direct homing and engraftment of HSPCs after transplantation to BM are still not well understood. We propose that these processes could be enhanced by employing some safe, innovative, and efficient approaches. We will shed more light on these processes and propose new innovative strategies by i) improving the responsiveness of infused HSPCs so that they can better migrate toward a gradient of the main BM chemoattractant, stromal-derived factor 1 (SDF-1), and the two supporting BM chemoattractants, sphingosine-1-phosphate (S1P) and extracellular adenosine triphosphate (eATP), and ii) we will manipulate in BM microenvironment of transplant recipient a state of “sterile inflammation” induced by myeloablative condition for transplantation, to promote better homing and engraftment. Our pioneering data indicate that receptors for HSPCs chemoattractants, including CXCR4 for SDF-1, S1P1R for S1P, and P2X7 for eATP, have to be included in membrane lipid rafts (MLRs) for optimal migration and BM homing. These MLRs are assembled on the cell surface in response to NADPH oxidase 2 (Nox2) that generates reactive oxygen species (ROS) to trigger activation of Nlrp3 inflammasome. Thus, we propose that the Nox2-ROS-Nlrp3 inflammasome axis regulates the migration of HSPCs and their homing properties. Similarly, our data indicate that the same axis facilitates the homing response of the BM microenvironment after myeloablative conditioning before transplantation. Therefore, based on our intriguing published and preliminary evidence, we propose three interrelated specific aims. Specific Aim 1. To enhance the Nox2–ROS–Nlrp3 inflammasome-mediated MLRs formation to increase migration of HSPCs to BM chemoattractants. We will test strategies to improve the responsiveness of transplanted HSPCs to BM chemoattractants by increasing the formation of MLRs on HSPCs in Nox2–ROS–Nlrp3 inflammasome-dependent manner. Specific Aim: To enhance homing of HSPCs by modulating Nox2-ROS-Nlrp3 inflammasome mediated sterile inflammation in the recipient BM microenvironment after myeloablative conditioning for transplantation. Since myeloablative conditioning for transplantation fuels a state of “sterile inflammation” in the BM microenvironment that directs the homing of transplanted HSPCs, we will manipulate the state of sterile inflammation in the BM microenvironment to promote better homing and engraftment. Specific Aim 3. To explain at the cellular and molecular level, the role of the Nox2-ROS-Nlrp3 inflammasome axis in migration, homing, and engraftment of HSPCs. We will employ state of the art strategies to analyze cellular and molecular effects that regulate i) optimal MLRs-dependent migration of HSPCs and ii) sterile inflammation induced BM microenvironment that facilitates homing and expansion of transplanted HSPCs.
Eligibility:
A suitable applicant should have the following qualifications:
- PhD degree in Biology or Biotechnology
- Basic previous experience in the following biology techniques: RNA seq, Mass Spectrometry, flow cytometry
- Academic background in cell biology, molecular biology, and/or genetics.
- Scientific research experience (full-text international publications, full-text articles published in Polish journals, international abstracts, active participation in (inter)national meetings, and scientific courses)
- Ability to work independently
- The candidate is required to have knowledge of stem cell biology
- Good knowledge of English
- Strong interest in science
How to apply:
Please send to:
mzrata01@louisville.edu
mariusz.ratajczak@wum.edu.pl
medycyna.regeneracyjna@wum.edu.pl
mariusz.ratajczak@louisville.edu and
henning@iq.usp.br
*Documents (as PDF files):
- Letter of interest
- CV
- Publication list
- Photo
- Contact details of 1-2 potential referees with recommendation letter
- Statement (see below)
The e-mail heading should be: “Post-doc – OPUS grant”.
Please, also provide the statement that you grant us a permission to process your personal details for the recruitment process:
“I hereby give consent for my personal data included in the job offer to be processed for the purposes of recruitment conducted by the Medical University of Warsaw located in Warsaw”.
The rules for the protection of personal data used by the Medical University of Warsaw:
- The administrator of personal data is the Medical University of Warsaw located in Warsaw, Żwirki i Wigury 61, 02-091 Warszawa,
- Contact to the Data Protection Officer - email address: iod@wum.edu.pl.
- Personal data will be processed in order to implement the recruitment process pursuant to art. 221 of the Labor Code, and in the case of providing a broader scope of data pursuant to art. 6 § 1a GDPR - consent expressed by the candidate.
- Access to personal data within the University's organizational structure shall only have employees authorized by the Administrator in the necessary scope.
- Personal data will not be disclosed to other entities, except for entities authorized by law.
- Personal data will be stored for the period necessary to carry out the recruitment process, up to 12 months from the settlement of the recruitment process. After this period, they will be removed.
- You have the right to access your data, the right to rectify, delete, limit processing, the right to transfer data, the right to object to the processing, the right to withdraw consent.
- You have the right to withdraw consent to the processing of your personal data at any time, which will not affect the lawfulness of the processing that was carried out on the basis of consent before its withdrawal.
- You have the right to lodge a complaint with the Office for Personal Data Protection when it is justified that his personal data are processed by the Administrator in breach of the general regulation on the protection of personal data of April 27, 2016.
- Providing personal data is voluntary, but necessary to participate in the recruitment process to the extent specified in art. 221 § 1 of the Labor Code, voluntary in the remaining scope.
- Decisions will not be taken in an automated manner and personal data will not be subject to profiling.

Saline solution inhibits SARS-CoV-2 replication
Along with professors Dr. Edson L. Durigon and Dr. Cristiane R. Guzzo, the group of prof. Henning demonstrated that, in lung epithelial cells, hypertonic saline inhibits the replication of the COVID-19 virus. This finding may help in the search for treatments and methods of prophylaxis of COVID-19.
Check out the articles published in Brazilian communication vehicles (in Portuguese):
Access the full article: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8442612/pdf/pt1c00080.pdf
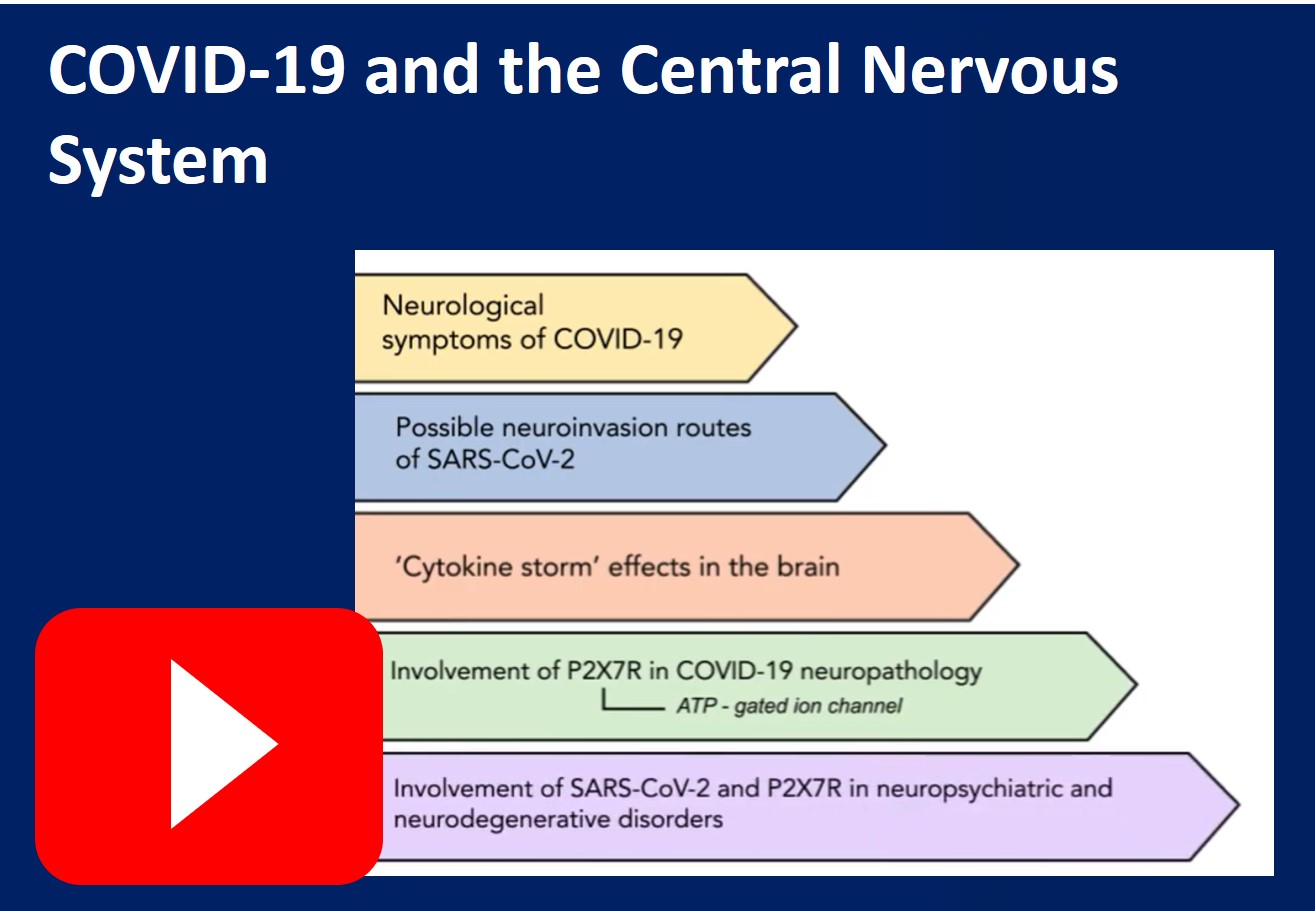
COVID-19 and the Central Nervous System
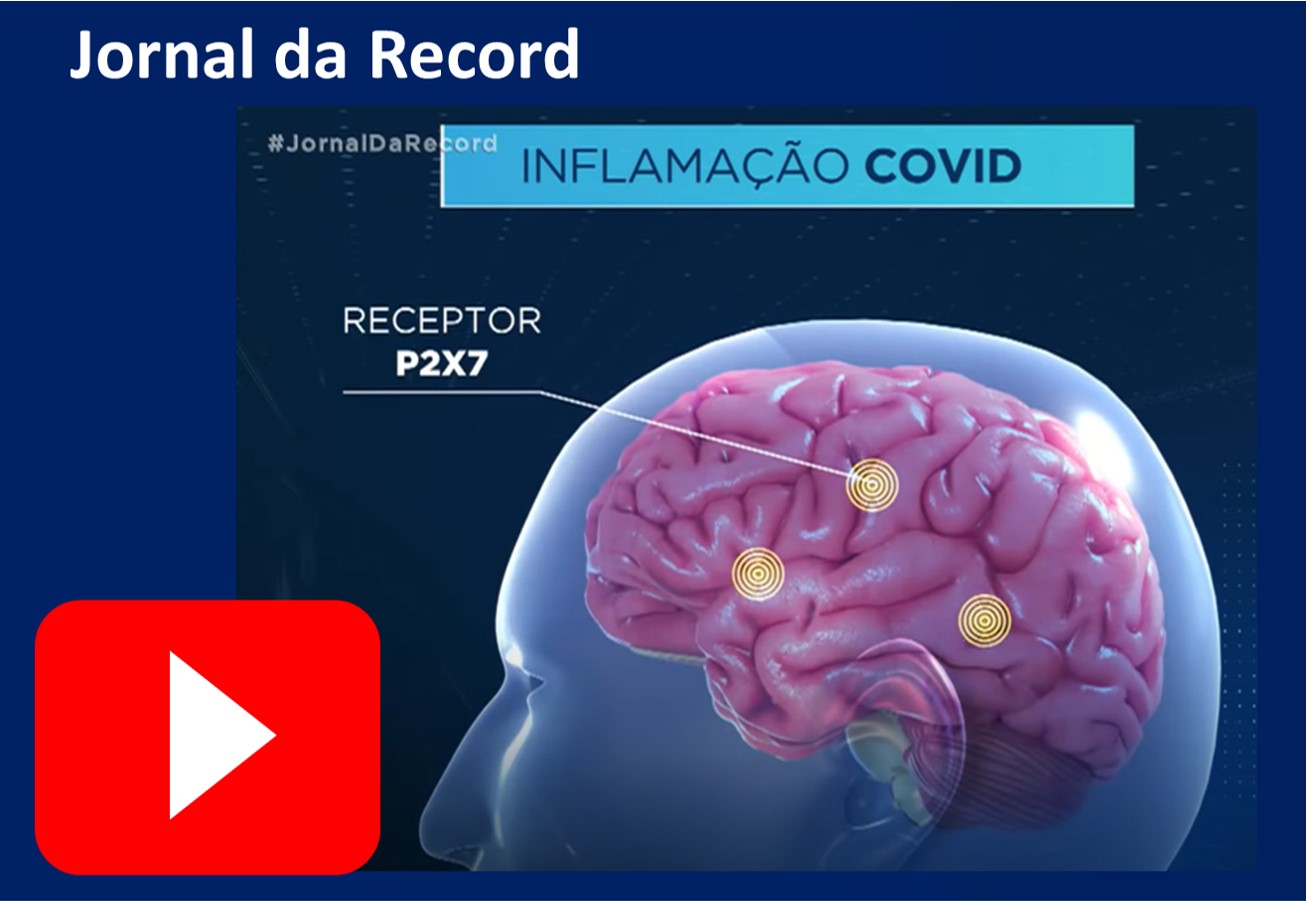
The research group of professor Dr. Henning Ulrich recently reviewed the possible mechanisms of the SARS-CoV-2 virus invasion of the Central Nervous System and proposed that these mechanisms involve ATP activation of the receptor P2X7. Thus, drugs that block this receptor should be a promising strategy to prevent or treat neurological complications in patients with COVID-19. Neurological symptoms are being reported in patients with COVID-19, but the acute and long-term consequences of SARS-CoV-2 on the central nervous system are still unknown.
ATP is known to be released in situations of inflammation and cell death. The severity of COVID-19 is associated with exacerbated inflammatory responses, causing the so-called “cytokine storm”. Cytokines are factors that trigger and worsen inflammation and, together with SARS-CoV-2, can reach the central nervous system, which can induce mini-strokes and neuron death. These processes are widely described in psychiatric disorders and neurodegenerative diseases. Dr. Ulrich and collaborators proposed that SARS-CoV-2 infection can trigger and / or worsen central nervous system diseases (such as depression, illness Parkinson's and Alzheimer's). The increase in ATP induced by SARS-CoV-2 infection can activate P2X7 receptors and worsen the inflammatory condition in these diseases. Additionally, patients with these diseases may be more susceptible to SARS-CoV-2 neuroinfection. Watch the video for more details.
This work was covered on the Brazilian television station Record, on the program Jornal da TV Record on March 27, 2021, and the video is available below (In portuguese).
Full article available at: https://www.nature.com/articles/s41380-020-00965-3
Recently published manuscript:
Rafael R. G. Machado, Talita Glaser, Danielle B. Araujo, Lyvia Lintzmaier Petiz, Danielle B. L. Oliveira, Giuliana S. Durigon, Alessandra L. Leal, João Renato R. Pinho, Luis C. S. Ferreira, Henning Ulrich*, Edison L. Durigon*, Cristiane Rodrigues Guzzo* (2021). Inhibition of Severe Acute Respiratory Syndrome Coronavirus 2 Replication by Hypertonic Saline Solution in Lung and Kidney Epithelial Cells. ACS Pharmacology & Translational Science: 10.1021/acsptsci.1c00080
Link: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8442612/pdf/pt1c00080.pdf
Deidiane Elisa Ribeiro, Ágatha Oliveira-Giacomelli, Talita Glaser, Vanessa F. Arnaud-Sampaio, Roberta Andrejew, Luiz Dieckmann, Juliana Baranova, Claudiana Lameu, Mariusz Z. Ratajczak & Henning Ulrich (2020). Hyperactivation of P2X7 receptors as a culprit of COVID-19 neuropathology. Molecular Psychiatry: https://doi.org/10.1038/s41380-020-00965-3.
Link: https://www.nature.com/articles/s41380-020-00965-3
Micheli Mainardi Pillat, Arne Krüger, Lara Mendes Ferreira Guimarães, Claudiana Lameu, Edmarcia Elisa de Souza, Carsten Wrenger, Henning Ulrich (2020). Insights in chloroquine action: perspectives and implications in Malaria and COVID‐19. Cytometry A. doi: 10.1002/cyto.a.24190
Link: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7404934/
Anacleto Silva de Souza, Jose David Rivera, Vitor Medeiros Almeida, Pingju Ge, Robson Francisco de Souza, Chuck Shaker Farah, Henning Ulrich, Sandro Roberto Marana, Roberto Kopke Salinas, Cristiane Rodrigues Guzzo (2020). Molecular Dynamics Reveals Complex Compensatory Effects of Ionic Strength on the Severe Acute Respiratory Syndrome Coronavirus 2 Spike/Human Angiotensin-Converting Enzyme 2 Interaction. J Phys Chem Lett. doi: 10.1021/acs.jpclett.0c02602
Link: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7737395/
Henning Ulrich and Micheli M. Pillat (2020). CD147 as a Target for COVID-19 Treatment: Suggested Effects of Azithromycin and Stem Cell Engagement. Stem Cell Reviews and Reports ://doi.org/10.1007/s12015-020-09976-7.
Link: https://link.springer.com/article/10.1007/s12015-020-09976-7
New projects:
Mechanism of cellular infection of the SARS-CoV-2 virus
In December 2019, one of the greatest challenges facing humanity today began in China: the pandemic COVID-19. COVID-19 is transmitted by the SARS-CoV-2 virus, characterized by causing severe acute respiratory syndrome due to impaired lung function. The lung of adult individuals has stem cells and endogenous progenitors, with a characteristic of multipotency and self-renewal. Although they remain quiescent in baseline conditions, these cells are important for epithelial cell homeostasis and injury repair. Recent studies suggest that, with the exception of hair cells, most pulmonary epithelial cells can acquire stem cell characteristics and induce pulmonary repair. Studies show that both stem cells and cells of the central nervous system can be infected with the SARS-CoV-2 virus. The loss of smell and taste characteristic of COVID-19 indicates that the virus can affect the central nervous system. In addition, some of the patients showed neurological symptoms, such as headache, vomiting and nausea. Cellular infection by the SARS-CoV-2 virus occurs through the interaction between a viral protein called Spike and two proteins located on the cell surface, the angiotensin-converting enzyme 2 (ECA2) and CD147. CD147 is widely expressed in migratory neural progenitor cells. According to studies, the overexpression of this protein in neural progenitor cells increased the cell migration from the subventricular zone to sites of ischemic injury, which suggests the participation of the CD147 protein in neuro-regenerative processes. Based on the evidence discussed, the objectives of the present project are to analyze (1) the expression of CD-147, its splicing variants and its level of glycosylation in differentiated iPSCs in pulmonary progenitor cells and infected neural progenitor cells; (2) whether the SARS-CoV-2 virus is capable of infecting pulmonary progenitor cells and neural progenitor cells; (3) the effects of viral infection on the proliferation and differentiation of these cells; (4) the effects induced by the inhibition of the CD-147 protein by antibodies on the cell invasion by the SARS-CoV-2 virus. In addition, we will study the interaction between the CD147 receptor and the viral spike protein by means of denaturation, isothermal calorimetry and nuclear magnetic resonance in solution, state-of-the-art technology for studying the structure and dynamics of proteins. The results obtained will allow a better understanding of the mechanism of cellular infection of SARS-CoV-2 as well as the study of a new strategy to inhibit it by blocking the CD147 protein.
Evaluation of the ATP-adenosine axis in lymphocytes in SARS-CoV-2 infection
Maria Notomi Sato, Anna Julia Pietrobon, Henning Ulrich, Talita Glaser and Ana Battastini.

Initial Deficit in Huntington's Disease
A work recently published led by Prof. Henning Ulrich (USP) contributes to a better understanding of Huntington's disease, a neurodegenerative disease. The research conducted by Prof. Dr. Henning Ulrich and Dr. Talita Glaser showed that P2X7 and P2Y2 receptors may induce the opening of voltage-dependent calcium channels, resulting in a possible impairment in the differentiation of neural stem cells in GABA-secreting neurons. Using the cell culture approach to study Huntington’s disease, the authors found that GABAergic neurons from neural stem cells present dysfunctional P2Y2 receptor, resulting in alterations of cellular homeostasis and higher susceptibility to neuron death. Additionally, using a pioneering strategy for real-time imaging of gene expression, this work showed how spontaneous calcium oscillations dynamically regulate neurogenesis. This work evidenced a new paradigm in Huntington’s field due to the early disease manifestations, resulting in a new point of view as a neurodevelopmental disease. Therefore, it was evidenced the importance of basic science and how it can impact on clinical knowledge. The work was mostly developed at IQ-USP, with collaborations at the University of Kyoto and Harvard Medical School.
Link for the article:
Purine and Kinin Receptors in Neurogenesis and Neurodegeneration
Neurogenesis originates the differentiation of neurons from neural stem cells (NSC). This process is principally taking place during embryonic development, but is also present throughout life in specific brain regions, such as the subventricular zone and the subgranular zone of the hippocampus, where it increases neuronal plasticity and also contributes to regeneration of neural tissue. In case of dysregulation of neurogenesis, pathological dysfunctions may develop. Therefore, studies of mechanisms regulating neurogenesis are not only important for understanding developmental origins of brain diseases, but also for the therapy of neurodegenerative diseases through cell therapy. Our laboratory has been leading with discoveries in this field, as for example the participation of purinergic and kinin signaling in neural differentiation of embryonic and neural stem cells in vitro as well as in mobilization of neural stem cells in vivo. Focus has been put on functions of purinergic receptors activated by ATP and other nucleotides, consisting of P2Y1,2,4,6,11,12,13,14 G protein-coupled subtypes and P2X1-7 ligand-gated ion channels, as well as on G protein-coupled kinin-B1 and B2 receptors activated by bradykinin, kallidin and their metabolites. We showed that defined expression and activity patterns of these receptors contribute to the progress of differentiation and neural phenotype determination. However, in addition to their importance in guiding neurogenesis and contributing to brain functioning, extracellular nucleotide- and kinin- induced signaling pathways may be targeted in order to revert proinflammatory processes and neurological pathologies leading to neurodegeneration, such as occurring during Parkinson’s, Huntington’s and Alzheimer’s disease and bipolar disorder. Below, we discuss some results of pharmacological activity modulation of purinergic kinin and purinergic receptors to provide mechanisms for Parkinson’s and Huntington’s disease treatment.
New Thematic Project:
Purine and bradykinin receptors as targets of study and therapeutic interventions in neurological diseases
The biological process of neurogenesis, that consists of differentiation of neural stem cells (NSC) into neurons, occurs both in embryonic and adult phases. Therefore, NSC are being proposed as interesting tools for nervous system tissue regeneration and restoration of physiological conditions, as well as models for elucidation of pathophysiological development of neurological diseases. Previous studies of our laboratory showed several signaling pathways involved in NSC development, especially through purinergic and bradykinin receptors in induction of differentiation and neural phenotype determination. These receptors and their signaling pathways also participate in neurodegenerative disorders, including Parkinson’s, Alzheimer’s and Huntington’s disease and autism, and are subject to targeting by specific pharmacological modulation of these pathways. In vitro and in vivo studies are coupled for determining the pharmacological and therapeutic efficiency of these drugs. Screening for neuroregenerative and neuroprotective effects are connected with immune status determination, as reduction of neuroinflammation has shown to be important for disease prevention and recovery. Moreover, new tools based on nanotechnology, as biopolymers conjugated to therapeutic compounds, are used for NSC transplantation improvement.
Publish in:



































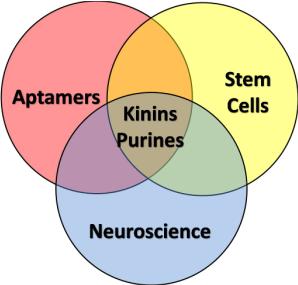
The Neuroscience Laboratory has several ongoing projects, involving three major areas: Aptamers, Stem Cells and Neuroscience. Furthermore, many projects are intended to elucidate the involvement of the purinergic and kininergic systems in diverse physiological and pathological processes. Click on the image to enlarge the abstracts.
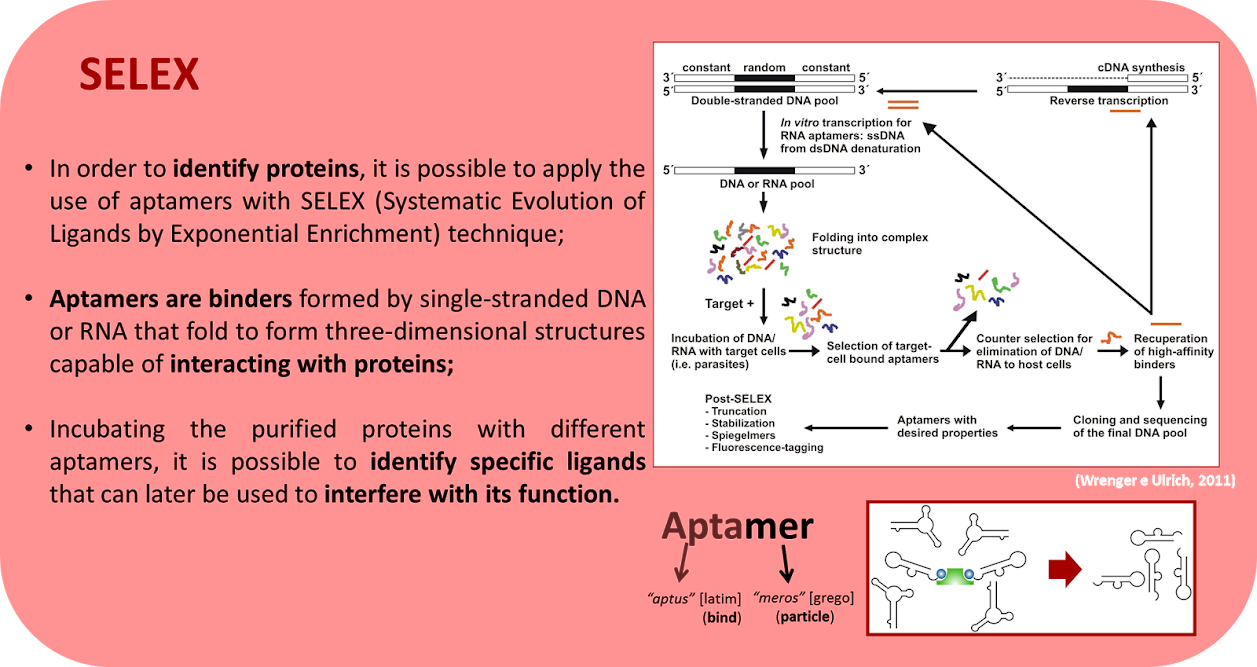
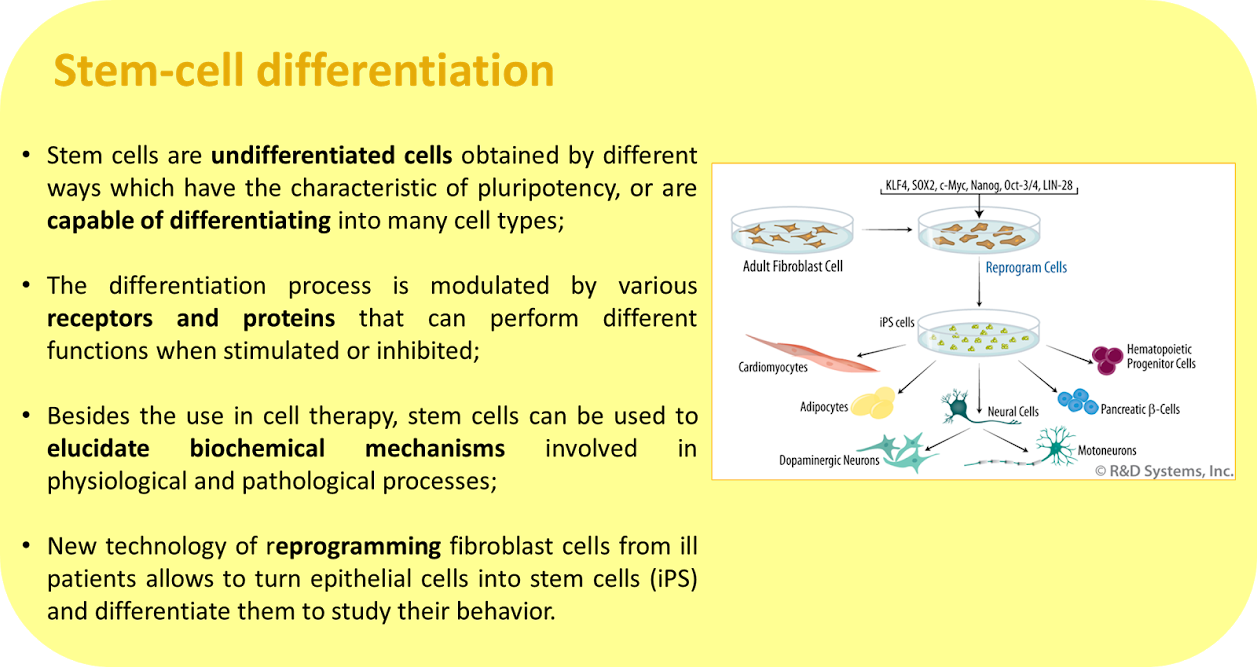
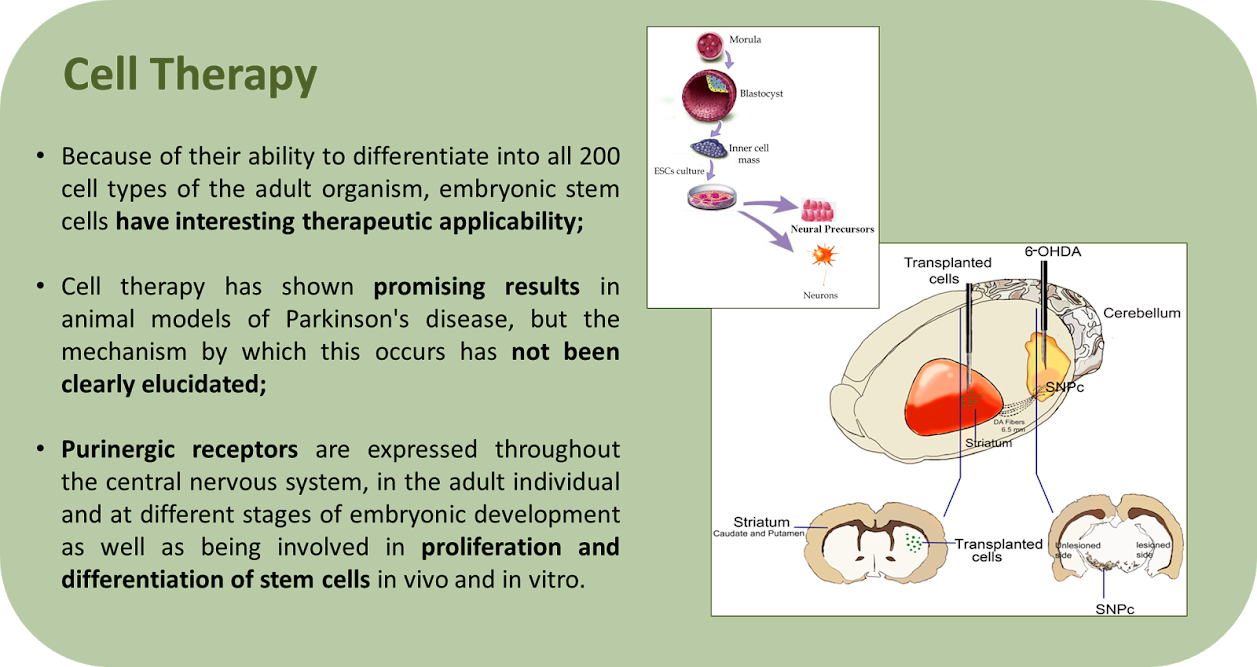

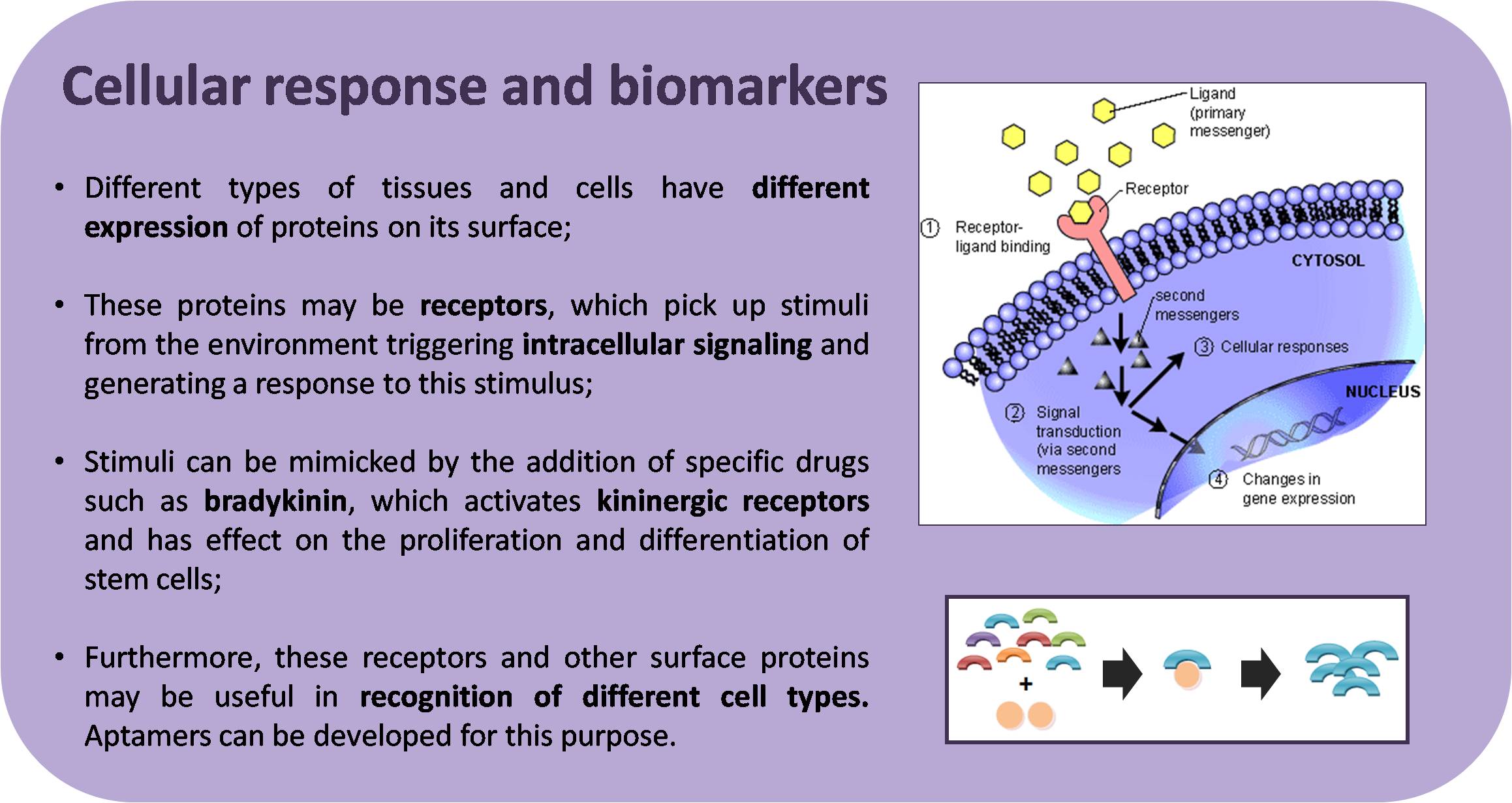

Coming soon.









 |
| Título |
Bases Moleculares da Biotecnologia |
| ISBN |
9788572417594 |
| Autores |
Ulrich, Henning; Colli, Walter; Faria, Marcella; Trujillo, Cleber A. |
| Selo Editorial |
ROCA |
| Edição |
1/2008 |
| Número de páginas |
232 |
|
|
A Engenharia Genética da década de 1970 inaugurou, de forma mais ampla, a moderna Biotecnologia com aplicação em áreas como Biologia e Medicina. Decorre desta concepção que a Biotecnologia é altamente interdisciplinar, sendo um dos campos de investigação científica que mais cresce e se desenvolve no mundo, razão pela qual Bases Moleculares da Biotecnologia pretende expor os estudantes e pesquisadores ao campo da biotecnologia molecular e ao potencial terapêutico de novas técnicas. São discutidos pontos-chave da transdução de sinal, que são alvo de tratamentos baseados em engenharia genética. A obra enfoca como se pode converter o progresso do conhecimento na biologia molecular em estratégias terapêuticas, a saber: técnicas para desenvolvimento racional de novos agentes terapêuticos, tais como oligonucleotídeos antisense, antigene e ácidos ribonucleicos de dupla fita para modular a expressão gênica; técnicas para desenvolvimento seletivo de peptídeos, proteínas e oligonucleotídeos isolados a partir de bibliotecas combinatórias e screening funcional. Também objetiva despertar o interesse dos estudantes pelas aplicações farmacêuticas e industriais resultantes da pesquisa fundamental em biomedicina, bem como abrir uma visão que possa levar a novas linhas de trabalho. Bases Moleculares da Biotecnologia focaliza o estado da arte em biotecnologia molecular, permitindo ao público uma análise crítica dos tópicos discutidos com característica inovadora no Instituto de Química e outros Institutos de grande prestígio da ciência brasileira, pois, ao se tratar do papel fundamental da Bioquímica e da Biologia Molecular na pesquisa dirigida à terapêutica e à indústria, é possível permitir a assimilação de conteúdos e ferramentas concretas para os interesses específicos de cada pesquisador e de cada estudante.
|
 |
| Título |
Perspectives of Stem Cells |
| ISBN |
9789048133758 |
| Editor |
Ulrich, Henning |
| Selo Editorial |
Springer |
| Edição |
1/2010 |
| Número de páginas |
285 |
|
|
Stem cells are fascinating cell types. They can replicate themselves forever while retaining the potential to generate progeny with specific functions. Because of these special properties, stem cells have been subjects of intensive investigation, from understanding basic mechanisms underlying tissue generation, to modeling human diseases, to application for cell replacement therapy. Stem cells come in different forms. For example, mouse embryonic stem cells can general all cell types in a body, either in a dish or when put back into mouse embryos. On the other hand, neural stem cells in the adult brain generate neurons and glia cells that contribute to the brain’s plasticity. Rapid progress has been made in the stem cell field with discoveries published in a record speed. A quick Pubmed search has returned 2789 hits for “embryonic stem cells” and 815 hits for “adult neural stem cells/neurogenesis” in the year 2008 alone. It remains a taunting task for all who are interested in stem cells to keep up with rapidly accumulating literatures. The “Perspectives of Stem Cells” by a truly international team of experts provides a timely and invaluable highlight of the stem cell field gearing toward future therapeutic applications in the nervous system. Stem cells with neural potentials have attracted a lot of attention because of their promise for cell replacement therapy, ranging from degenerative neurological disorders to spinal cord injuries.
|
 |
| Título |
Trends in Stem Cell Proliferation and Cancer Research |
| ISBN |
9789400762114 |
| Autores |
Resende, Rodrigo, R.; Ulrich, Henning. |
| Selo Editorial |
Springer |
| Edição |
1/2013 |
| Número de páginas |
661 |
|
|
The book will discuss the molecular mechanisms of cancer diseases, stem cell proliferation and transformation into cancer cells beyond the physiological processes that occur in normal stem cell biology. Some of the key oncogenic events in cancer and their signaling pathways that regulate cell division cycle progression will be described considering prospects for using such knowledge in advanced cancer therapy. Each chapter shall provide an invaluable resource for information on the most current advances in the field, with discussion of controversial issues and areas of emerging importance
|


|
www.clubedepurinas.com.br
|
|
The Brazilian Purine Club was founded in 2009 with the objective of bringing together the scientific community interested in topics related to purinergic signaling. The organization of periodical congresses aims to promote the integration of researchers and students into a scientific field, which is rapidly expanding in Brazil and worldwide. Based on that, four scientific congresses were held so far.
The “First Meeting of the Brazilian Purine Club: Purinergic signaling and therapeutic implications”, realized in August 2010 in Águas de Lindóia in São Paulo State, was attended by 150 participants (Researchers from Brazil and abroad in addition to a significant number of graduate and undergraduate students). The “Second Meeting of the Brazilian Purine Club” was realized in August 2011 in the city of Rio de Janeiro. The conference was attended by established researchers and students. These two initial meetings were organized as satellite symposiums of the annual conference of FESBE (Brazilian Federation of Experimental Biology Societies). The apparent success of the first two Brazilian Purine Club Meetings, demonstrated by the number of participants and increased scientific interactions between the members, encouraged us to organize a larger meeting in 2012. The “Third Meeting of the Brazilian Purine Club” was held in September in the town of Ouro Preto in the Minas Gerais State, followed in September 2013 by the ” Fourth Meeting of the Brazilian Purine Club” in Canela, Rio Grande do Sul. The latter congresses were organized and held independent from FESBE. Renowned scientists in the field of purinergic signaling from Brazil and abroad contributed to these meetings.
|
_______________________________________________________________________________________________________________________
Neuroscience Laboratory
Instituto de Química - IQ
Universidade de São Paulo - USP
Av. Prof. Lineu Prestes, 748, Butantã
CEP: 05508-000 - São Paulo - SP - Brasil
Building 9S, room 964
Telephone number (11) 3091-9181
Contact: henning@iq.usp.br
